In the world of semiconductors and advanced ceramics, if silicon was the "King of Steel" that dominated the 20th century, then Silicon Carbide is the "super-alloy" now rising to pioneer a new era. This material, hailed as a "Third-Generation Semi" star, did not appear out of nowhere. It has a long history spanning over a century, and its journey to commercialization reads like a tech industry "hero's origin" story.
Chapter 1: The "Hero's Origin" of SiC — A Century of Foundation, A Decade of Bloom
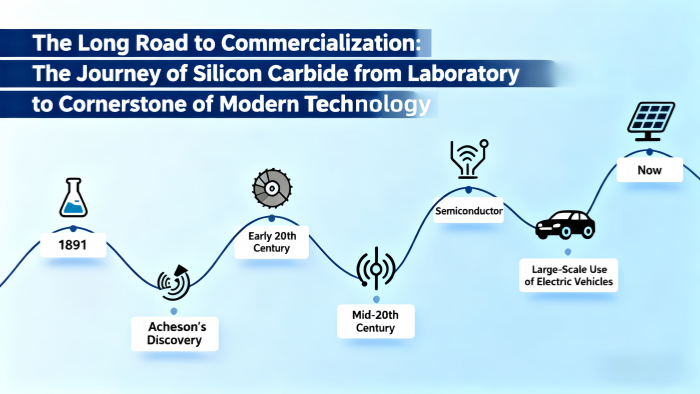
The discovery of Silicon Carbide is much older than we might think.
1891: The Accidental "Man-Made Star"
American inventor Edward Goodrich Acheson was attempting to synthesize artificial diamonds by heating a mixture of clay (aluminosilicate) and coke (carbon) with an electric current. He accidentally produced shiny, extremely hard crystals. Believing it to be a compound of carbon and corundum, he named it "Carborundum." This beautiful misunderstanding officially began the history of SiC.Early 20th Century: First Signs of Promise
Before the semiconductor age began, SiC was primarily used as an abrasive, thanks to its extreme hardness—the "grit" in sandpaper and cutting wheels.Mid-to-Late 20th Century: The Theoretical Prophet
Scientists long recognized that SiC possessed excellent semiconductor properties—a wide bandgap. In theory, it was far superior to silicon for creating high-performance, high-voltage, and high-temperature-resistant devices. However, theory was one thing; reality was another.
Why Did It Take So Long? — The "Extreme" Manufacturing Challenges
Transforming SiC from an excellent abrasive into a single-crystal substrate for electronics or a dense ceramic for structural components was immensely difficult. This formed the core barrier to its commercialization:
Difficult to "Grow": The Extreme Challenge of Crystal Growth
Silicon can be pulled like crystallizing sugar syrup to form large, pure, defect-free single crystals.
Silicon Carbide has no melting point; at atmospheric pressure, it sublimes directly (solid to gas). This meant it could not be grown using traditional melt-based methods like the Czochralski process.
The Solution: It wasn't until 1978 that Soviet scientist Tairov developed the Physical Vapor Transport (PVT) method, providing a viable path for growing SiC single crystals. However, this process was notoriously slow, energy-intensive, and difficult to control defect formation, resulting in early SiC substrates that were small, low-quality, and priced like gold.
Difficult to "Carve": Machining Hardness Rivaling Diamond
The hardness of SiC is second only to diamond. This meant that cutting, grinding, and polishing it were exceptionally difficult, leading to high processing costs and low yields.Difficult to "Densify": The Technical Hurdle of Ceramic Sintering
For Silicon Carbide ceramics (distinct from single-crystal SiC wafers for semiconductors), manufacturing was equally challenging. Pure SiC is bonded by covalent bonds with a very low diffusion coefficient, making it nearly impossible to densify through conventional sintering. This challenge spurred different technological paths, giving rise to the Silicon Carbide family we see today.
Chapter 2: The "Four Pillars" of the Silicon Carbide Family
In order to overcome the sintering problem, engineers have developed several mainstream processes, forming the core family of silicon carbide ceramics:
Reaction-Bonded Silicon Carbide (RBSiC)
The "Shortcut" Ingenuity: A mixture of SiC powder and carbon powder is formed into a shape and then reacted with molten silicon at high temperatures. The silicon infiltrates the pores, reacting with carbon to form new SiC and filling the remaining space.
Advantages: Lower sintering temperature, lower cost, ability to produce complex shapes.
Disadvantages: Contains residual free silicon, leading to reduced high-temperature performance (>1350°C) and slightly inferior resistance to strong alkalis.
Pressureless Sintered Silicon Carbide (SiSiC)
The "Hardcore" Tech Champion: Densification is achieved at very high temperatures using sintering aids, without applying external pressure.
Advantages: Top-tier purity, density, strength, hardness, corrosion resistance, and high-temperature stability. Considered the performance "king."
Disadvantages: High requirements for raw material powder, difficult process, higher cost.
Recrystallized Silicon Carbide (RSiC)
The Pinnacle of "Purity": A special type of pressureless sintering performed at extremely high temperatures without any additives, relying solely on evaporation-condensation between SiC particle surfaces for bonding.
Advantages: Extremely high purity, excellent thermal shock resistance and high-temperature load-bearing capacity. Ideal for high-grade kiln furniture (e.g., rollers, beams).
Disadvantages: Contains some closed porosity, room-temperature strength is slightly lower than Pressureless Sintered SiC.
Silicon Nitride Bonded Silicon Carbide (NSiC)
The "Power Couple" Exemplar: Uses SiC as the aggregate, with reacted Silicon Nitride acting as the bonding phase.
Advantages: Perfectly combines the thermal conductivity and wear resistance of SiC with the strength and toughness of Silicon Nitride, offering outstanding thermal shock resistance.
Disadvantages: The Silicon Nitride bond phase can be compromised in strongly oxidizing atmospheres.
Chapter 3: Breaking the Cocoon — Why Now?
The commercial explosion of SiC is the result of several converging factors:
Demand Pull: Industries like electric vehicles, renewable energy, and 5G created a "surge" in demand for high-efficiency, compact power devices. The performance ceiling of silicon-based devices had become a bottleneck, and the market was calling for a "savior."
Process Maturation: After decades of R&D, the yield and cost of growing larger-diameter substrates via the PVT method (from 2-inch to today's 6-inch and 8-inch) have improved significantly. Ceramic processes like Pressureless Sintering have also achieved stable, scaled production.
Supply Chain Formation: A complete global supply chain—from substrates and epitaxy to device design, fabrication, and module packaging—has formed and matured, driving rapid technological iteration and continuous cost reduction.
Conclusion
From a chance discovery in a laboratory in 1891 to now bearing the responsibility of an energy revolution, the century-long journey of Silicon Carbide exemplifies the meaning of "success comes to those who prepare." It is no longer just an abrasive trapped in sandpaper. It has transformed into the "heart" powering electric vehicles, the "manager" boosting solar efficiency, and the "backbone" supporting industrial energy savings. As costs continue to fall and processes keep refining, the Silicon Carbide family is destined to write an even more brilliant chapter in this new era of electrification.
[Contact us to Inquire or Order] or [Call Our Hotline].
We can also accommodate your needs even for small quantity orders.Click Here to Visit Our Official Retail Site and Explore Product Details Now!——kilnshelves-sic.com











